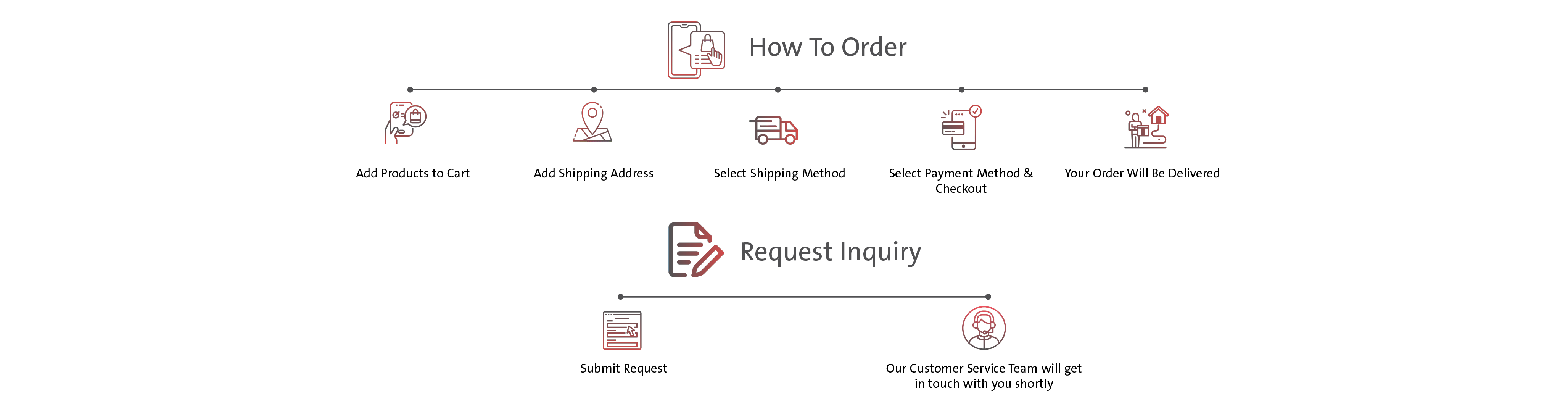



Introduction
Rubella (commonly known as German measles or 3-day measles) is an infection that primarily affects the skin and lymph nodes. It is caused by the rubella virus (not the same virus that causes measles), which is usually transmitted by secretions from the nose or throat. It can also pass through a pregnant woman's bloodstream to infect her unborn child. As this is a generally mild disease in children, the primary medical danger of rubella is the infection of pregnant women, which may cause congenital rubella syndrome in developing babies.
Intended Use
RubellaIgG/IgM Rapid Test is a solid phase immunochromatographic assay for the rapid, qualitative, and differential detection of IgG and IgM antibodies to Rubella virus in human serum plasma or whole blood. This test provides only a preliminary test result. Therefore, more specific alternative diagnosis methods such as the Sabin-Feldman dye test for IgG antibodies, the IgM-IFA (Immuno-fluorescent Antibody) test, and the PCR test must be used in order to obtain a confirmation of Rubella infection.
Principle
Rubella IgG/IgM test device has 3 pre-coated lines, “G” (Rubella IgG Test Line), “M” (Rubella IgM Test Line), and “C” (Control Line) on the surface of the membrane. All three lines in the result window are not visible before applying any samples. The “Control Line” is used for procedural control. The Control line should always appear if the test procedure is performed properly and the test reagents of the control line are working. A purple “G” and “M” lines will be visible in the result window if there are enough IgG and/or IgM antibodies to the Rubella virus in the sample. If IgG and/or IgM antibodies to Rubella virus are not present in the sample, there is no color appearance in “G” and/or “M”
| Product More Specifications | |
| Made In | USA |
- Stock: Pre Order
- Model: Acro Biotech Inc
- SKU: IRGM-302